DEVA Holding Çerkezköy-I Manufacturing Facility has an enclosed area of 43,000 m2 established on an area of 52,000 m2 in the Çerkezköy Organized Industrial Zone, at a distance of approximately 110 km from Istanbul.
The facility is made up of modern buildings where liquids/semi-solids and solids, cephalosporin, penicillin, hormone products, inhaled products, sterile liquid ampoules and vials and soft gel capsules are manufactured.
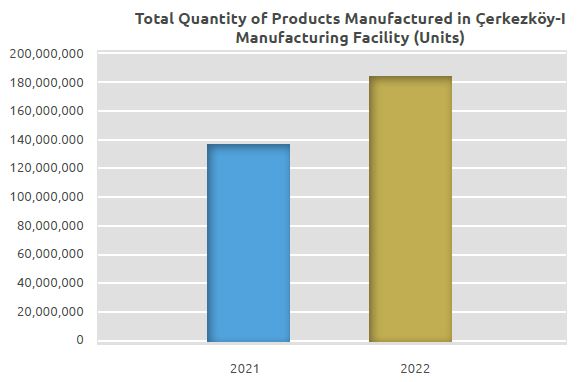
While a total of 137 million units were manufactured in the manufacturing facility in 2021, 183 million units were manufactured in 2022. The number of units manufactured in 2022 increased by 46 million units compared to 2021, which corresponds to an increase of 34%.
Liquids/Semi-Solids and Solids Manufacturing Unit
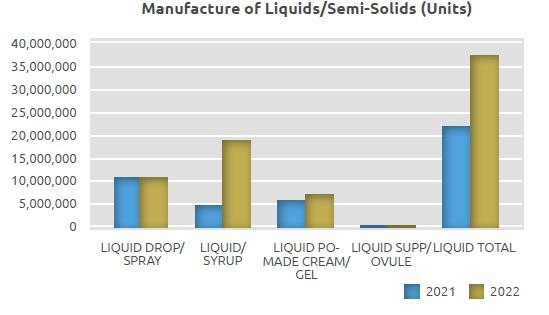
In the Liquids/Semi-Solids Manufacturing Unit, medicines in the form of syrup, drops and spray, cream and gel, suppositories and ovules are manufactured with state-ofthe-art technology.
In this unit, where a total of 22 million units of products were manufactured in 2021, a total of 37.5 million units, including 30 million units of syrups, drops and sprays and 7.5 million units of pomades, gels, suppositories and ovules, were manufactured in 2022.
The quantity manufactured in the Liquids Manufacturing Unit in 2022 on a unit basis increased by 70% compared to 2021, with 15.5 million more units manufactured.

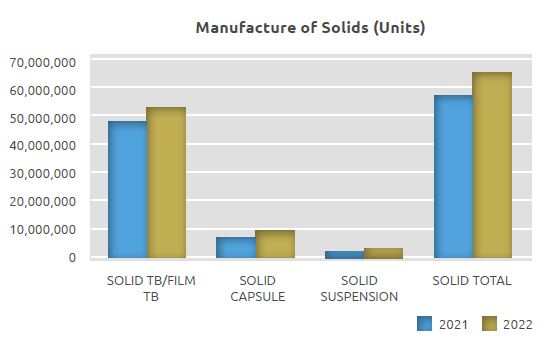
In the Solids Manufacturing Unit, medicines in the form of tablets/film tablets, capsules, and powders for suspension are manufactured with state-of-the-art technology.
In this unit, where 57.9 million units of products were manufactured in 2021, a total of 65.9 million units of products were manufactured in 2022, including 62.7 million units of tablets/capsules and soft gel capsules, and 3.2 million units of suspensions.
The quantity manufactured in the Solids Manufacturing Unit in 2022 on a unit basis increased by 14% compared to 2021, with 8 million more units manufactured.

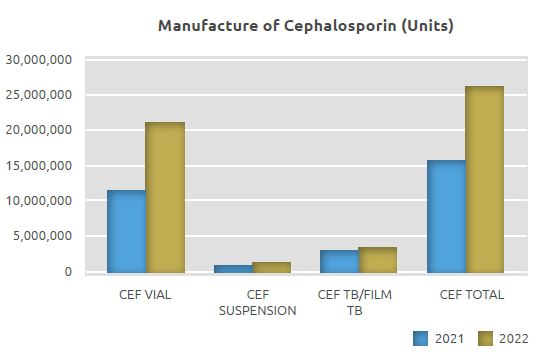
Cephalosporin Manufacturing Unit (Beta-lactam I)
While 15.8 million units were manufactured in the Cephalosporin Manufacturing Unit in 2021, 21.3 million units of sterile powder vials for injection, 1.6 million units of oral suspension, 3.4 million units of film coated tablets and capsules, totaling 26.3 million units, were manufactured in 2022.
The quantity manufactured in the Cephalosporin Manufacturing Unit in 2022 in terms of units increased by 66% compared to 2021, with 10.5 million more units manufactured.

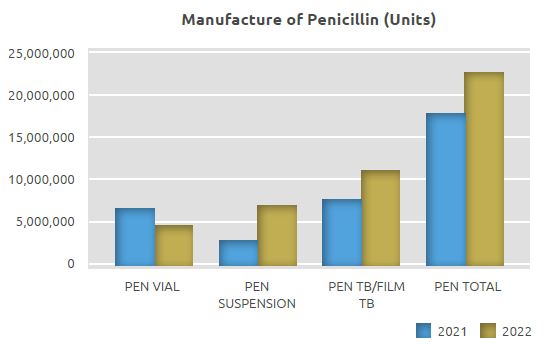
Penicillin Manufacturing Unit (Beta-lactam II)
While 17.6 million units were manufactured in the Penicillin Manufacturing Unit in 2021, 4.7 million units of sterile powder vials for injection, 7 million units of oral suspension, 10.9 million units of film coated tablets, totaling 22.6 million units, were manufactured in 2022.
The quantity manufactured in the Penicillin Manufacturing Unit in 2022 on a unit basis increased by 28% compared to 2021, with 5 million more units manufactured.

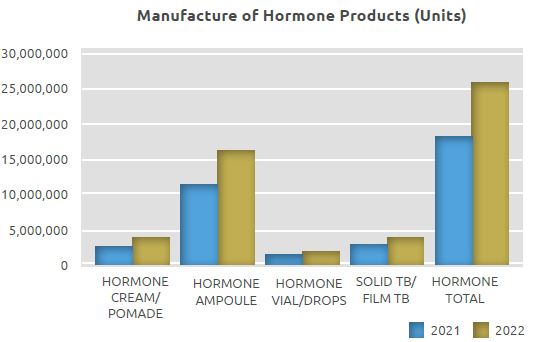
Hormone Products Manufacturing Unit
While 18.2 million units were manufactured in the Hormone Products Manufacturing Unit in 2021, 4 million units of cream/pomade, 4 million units of solid tablets and film tablets, and 17.9 million units of sterile ampoules, vials and drops, totaling 25.9 million units, were manufactured in 2022.
The quantity manufactured in the Hormone Products Manufacturing Unit in 2022 on a unit basis increased by 42% compared to 2021, with 7.7 million more units manufactured.

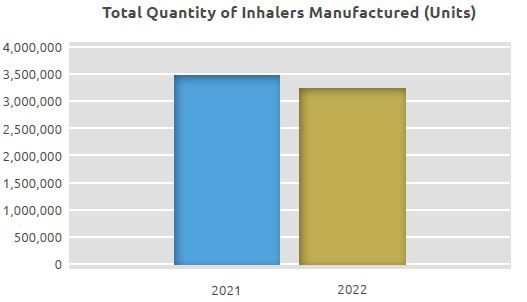
Inhaled Products Manufacturing Area
While 3.5 million units of MDIs and DPIs were manufactured in the Inhaled Products Manufacturing Unit in 2021, 3.3 million units were manufactured in 2022.

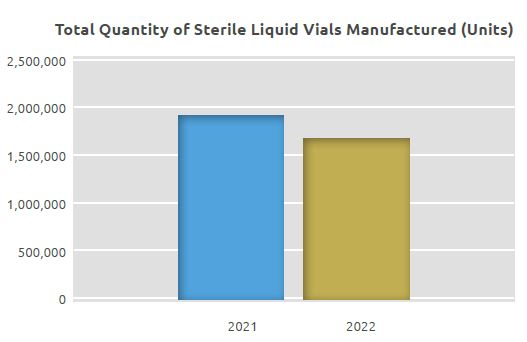
Sterile Liquid Ampoules, Vials and Soft Gel Capsules Manufacturing Unit
While 1.9 million units of sterile vials were manufactured in the Sterile Liquid Ampoules, Vials and Soft Gel Capsules Manufacturing Unit in 2021, 1.7 million units of sterile vials and 3.1 million solvent ampoules were manufactured in 2022.