
DEVA Holding Kartepe Manufacturing Facilities, located by the İzmit - Ankara highway, in Kartepe, Kocaeli, established on an area of 32,000 m² with an enclosed area of 16,500 m² is made up of modern buildings that manufacture sterile liquid drugs, empty medical ampoules and colognes.
Manufacture of Sterile Liquid Drugs
At Kartepe Sterile Liquid Drug Manufacturing Unit, sterile liquid ampoules for injection, sterile liquid vials for injection, sterile lyophilized liquid vials for injection, sterile liquid eye drops in ready-to-use PE bottles manufactured with the BFS (Blow-Fill-Seal) technology, sterile liquid solvent vials and vials containing inhalation solution for sterile nebulization are manufactured.
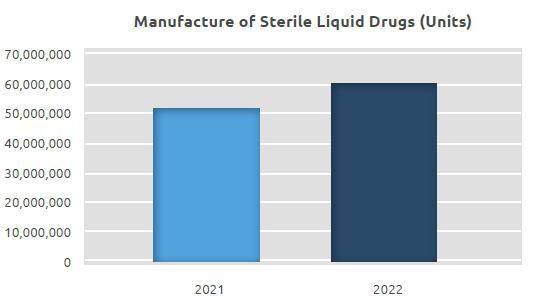
At the facility, where a total of 51.6 million units of sterile liquid drugs were manufactured in 2021, a total of 60.6 million units were manufactured in 2022, with an increase of approximately 17.5% compared to 2021.

Manufacture of Sterile Ampoules for Injection
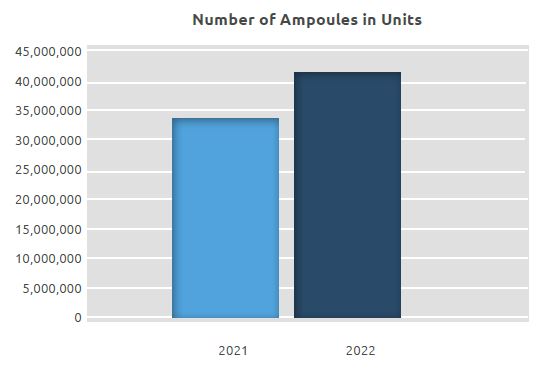
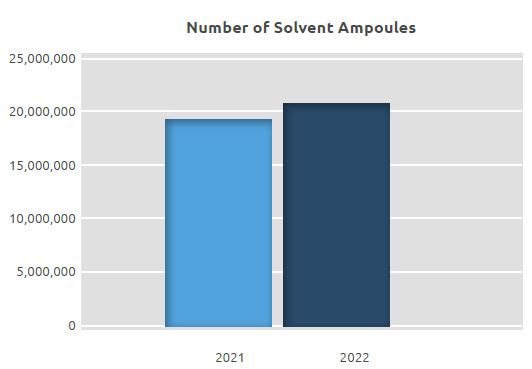
At the facility, where a total of 33.8 million units of ampoules and 19.6 million solvent ampoules were manufactured in 2021, a total of 41.4 million units of ampoules and 21.2 million units of solvents were manufactured in 2022. An increase of approximately 23% was seen in manufacture on a unit basis compared to 2021.

Manufacture of Sterile Liquid Vials for Injection and Sterile Liquid Lyophilized Vials for Injection
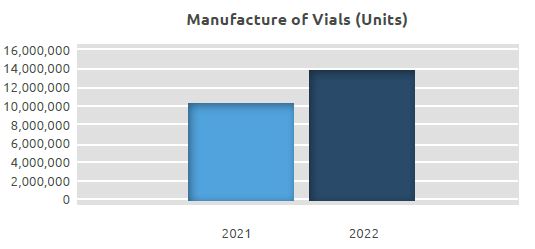
At our production line of Sterile Liquid Vials for Injection and Sterile Lyophilized Liquid Vials for Injection, where a total of 10.27 million units of vials were manufactured in 2021, a total of 13.84 million units of vials were manufactured in 2022. An increase of approximately 35% was seen in manufacture compared to 2021.

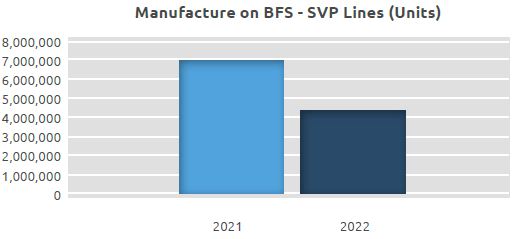
Manufacture of BFS-SVP
At our BFS-SVP production lines, where a total of 7.02 million units were manufactured in 2021, a total of 4.35 million units were manufactured in 2022.

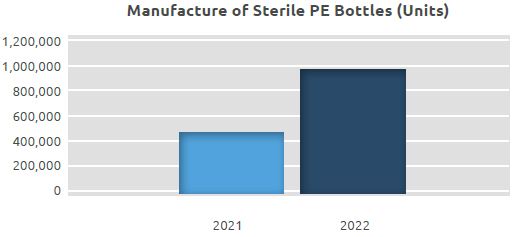
Manufacture of Sterile PE Bottles
In our filling line of eye drops in sterile PE bottles, where 0.47 million units were manufactured in 2021, a total of 0.97 million units were manufactured in 2022.

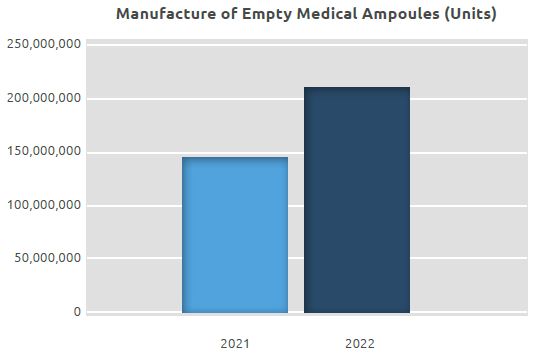
Manufacture of Empty Medical Ampoules
Our empty medical ampoule manufacturing unit meets a substantial part of the market’s need as one of the three leading medical ampoule manufacturing companies in our country.
Our unit, which also has an ISO 9001:2015 certificate, is inspected periodically by TUV Thuringen.
While the number of empty ampoules we manufactured in 2021 was 145.75 million, it was 209.80 million in 2022, seeing an increase of approximately 44% compared to 2021.

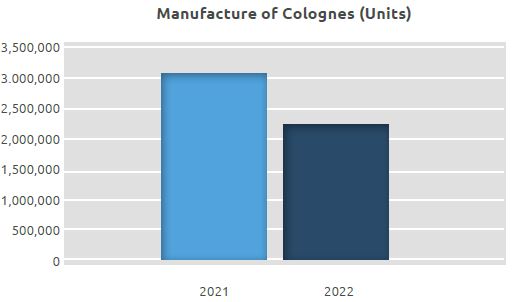
Manufacture of Cologne
Boğaziçi Kolonyaları, one of Türkiye’s best-selling brand, is produced in line with the Turkish Ministry of Health, Cosmetic GMP practices.
Our unit, which holds an ISO 9001:2015 certificate, is also inspected periodically by TUV Thuringen.
While the number of colognes we manufactured in 2021 was 3.06 million, it was 2.23 million in 2022.
The total capacity utilization rate of the Kartepe Manufacturing Facilities is 87.6%.

PRODUCTION INDICATORS
The total human and veterinary products manufactured in our manufacturing facilities on a unit basis was 200.7 million units in 2019, 205.8 million units in 2020, 194.5 million units in 2021 and 247.8 million units in 2022.
The total manufacturing value (drugs and other products) was realized as TRY 687.6 million in 2019, TRY 868.8 million in 2020, TRY 1,073.7 million in 2021 and TRY 2,199.3 million in 2022.